inDrop#
Check this GitHub page to see how inDrop libraries are generated experimentally. This is a droplet-based method, where cells are captured inside droplets. At the same time, gel beads with barcoded oligo-dT primer containing UMIs are also captured inside the droplet. Reverse transcription happens inside the droplet. The cells and gel beads are loaded on the microfluidic device at certain concentrations, such that a fraction of droplets contain only one cell AND one bead. The barcodes on the gel beads are generated using a split-pool based combinatorial indexing strategy.
Important
Be aware that there are different versions of inDrop. There is also a commercial version from 1CellBio. They have different adaptor sequences and final library structures. Therefore, they require different parameters for the preprocessing steps. Check the inDrop GitHub Page and the inDrop GitHub repository for some details.
The real situation is more complicated. If you do the experiments by yourself, you probably know the sequences. If you use a commercial inDrop platform or analyse published data, make sure you check the kit manual or the authors of the publication about the details of the sequences used in the protocol.
Some helpful threads: STARsolo issue #605, STARsolo issue #785 and indrops issue #32
If you find errors, which is highly likely, please do let me know via email
chenx9@sustech.edu.cnor raised an issue. Thank you in advance.
For Your Own Experiments#
The read configuration is a bit more complicated and depends on the version.
The V1 Configuration#
Order |
Read |
Cycle |
Description |
|---|---|---|---|
1 |
Read 1 |
>=47 |
This yields |
2 |
Index 1 (i7) |
6 or 8 |
This yields |
3 |
Index 2 (i5) |
Optional |
This yields |
4 |
Read 2 |
>=35 |
This yields |
The content of Read 1 is like this:
Length |
Sequence (5’ -> 3’) |
|---|---|
>46 |
8 - 11 bp + Barcode1 GAGTGATTGCTTGTGACGCCTT + 8 bp Barcode2 + 6 bp UMI + poly-T |
The V2 Configuration#
Order |
Read |
Cycle |
Description |
|---|---|---|---|
1 |
Read 1 |
>=35 |
This yields |
2 |
Index 1 (i7) |
6 or 8 |
This yields |
3 |
Index 2 (i5) |
Optional |
This yields |
4 |
Read 2 |
>=47 |
This yields |
The content of Read 2 is like this:
Length |
Sequence (5’ -> 3’) |
|---|---|
>46 |
8 - 11 bp + Barcode1 GAGTGATTGCTTGTGACGCCTT + 8 bp Barcode2 + 6 bp UMI + poly-T |
The V3 Configuration#
This configuration is more complicated and the naming of the output files does not really follow our normal convention. DO NOT get confused.
Order |
Read |
Cycle |
Description |
|---|---|---|---|
1 |
Read 1 |
>50 |
This normally yields |
2 |
Index 1 |
8 |
This normally yields |
3 |
Index 2 (i7) |
6 or 8 |
This normally yields |
4 |
Read 2 |
14 |
This normally yields |
The content of Read 2 is like this:
Length |
Sequence (5’ -> 3’) |
|---|---|
14 |
8 bp Barcode2 + 6 bp UMI |
You can think of the combination of 8 - 11 bp Barcode1 and 8 bp Barcode2 as the cell barcodes. If you use this method, you have to sequence the library on your own, you need to get the fastq files by running bcl2fastq by yourself. In this case it is better to write a SampleSheet.csv with i7 indices for each sample. Note that the i7 index is the 2nd read (Index 1) in V1 and V2, but the 3rd read (Index 2) in V3, so you need to adjust that accordingly. Here are examples of SampleSheet.csv of NextSeq runs with two samples using some standard index:
The V1 & V2 SampleSheet#
[Header],,,,,,,,,,,
IEMFileVersion,5,,,,,,,,,,
Date,17/12/2019,,,,,,,,,,
Workflow,GenerateFASTQ,,,,,,,,,,
Application,NextSeq FASTQ Only,,,,,,,,,,
Instrument Type,NextSeq/MiniSeq,,,,,,,,,,
Assay,AmpliSeq Library PLUS for Illumina,,,,,,,,,,
Index Adapters,AmpliSeq CD Indexes (384),,,,,,,,,,
Chemistry,Amplicon,,,,,,,,,,
,,,,,,,,,,,
[Reads],,,,,,,,,,,
50,,,,,,,,,,,
50,,,,,,,,,,,
,,,,,,,,,,,
[Settings],,,,,,,,,,,
,,,,,,,,,,,
[Data],,,,,,,,,,,
Sample_ID,Sample_Name,Sample_Plate,Sample_Well,Index_Plate,Index_Plate_Well,I7_Index_ID,index,I5_Index_ID,index2,Sample_Project,Description
Sample01,,,,,,BC1,CAGATC,,,,
Sample02,,,,,,BC2,ACTTGA,,,,
The V3 SampleSheet#
[Header],,,,,,,,,,,
IEMFileVersion,5,,,,,,,,,,
Date,17/12/2019,,,,,,,,,,
Workflow,GenerateFASTQ,,,,,,,,,,
Application,NextSeq FASTQ Only,,,,,,,,,,
Instrument Type,NextSeq/MiniSeq,,,,,,,,,,
Assay,AmpliSeq Library PLUS for Illumina,,,,,,,,,,
Index Adapters,AmpliSeq CD Indexes (384),,,,,,,,,,
Chemistry,Amplicon,,,,,,,,,,
,,,,,,,,,,,
[Reads],,,,,,,,,,,
50,,,,,,,,,,,
14,,,,,,,,,,,
,,,,,,,,,,,
[Settings],,,,,,,,,,,
,,,,,,,,,,,
[Data],,,,,,,,,,,
Sample_ID,Sample_Name,Sample_Plate,Sample_Well,Index_Plate,Index_Plate_Well,I7_Index_ID,index,I5_Index_ID,index2,Sample_Project,Description
Sample01,,,,,,BC1,CAGATC,,,,
Sample02,,,,,,BC2,ACTTGA,,,,
You need to run bcl2fastq differently based on the configuration like this:
# for V1 and V2 configuration
bcl2fastq --no-lane-splitting \
--ignore-missing-positions \
--ignore-missing-controls \
--ignore-missing-filter \
--ignore-missing-bcls \
-r 4 -w 4 -p 4
# for V3 configuration
bcl2fastq --use-bases-mask=Y50,Y8,I6,Y14 \
--create-fastq-for-index-reads \
--no-lane-splitting \
--ignore-missing-positions \
--ignore-missing-controls \
--ignore-missing-filter \
--ignore-missing-bcls \
-r 4 -w 4 -p 4
You can check the bcl2fastq manual for more information, but the important bit that needs explanation is --use-bases-mask=Y50,Y8,I6,Y14 in the V3 configuration. We have four reads, and that parameter specify how we treat each read in the stated order:
Y50at the first position indicates “use the cycle as a real read”, so you will get 50-nt sequences, output asR1_001.fastq.gz, because this is the 1st real read.Y8at the second position indicates “use the cycle as a real read”, so you will get 8-nt sequences, output asR2_001.fastq.gz, because this is the 2nd real read.I6at the third position indicates “use the cycle as an index read”, so you will get 6-nt sequences, output asI1_001.fastq.gz, because this is the 1st index read, though it is the 3rd read overall.Y14at the fourth position indicates “use the cycle as a real read”, so you will get 14-nt sequences, output asR3_001.fastq.gz, because this is the 3rd real read, though it is the 4th read overall.
After that, you will get two files per sample in the V1 and V2 configurations, and four files per sample in the V3 configuration:
# V1 configuration
Sample01_S1_R1_001.fastq.gz # 50 bp: barcodes, UMI and linkers, poly-T
Sample01_S1_R2_001.fastq.gz # 50 bp: cDNA reads
Sample02_S2_R1_001.fastq.gz # 50 bp: barcodes, UMI and linkers, poly-T
Sample02_S2_R2_001.fastq.gz # 50 bp: cDNA reads
# V2 configuration
Sample01_S1_R1_001.fastq.gz # 50 bp: cDNA reads
Sample01_S1_R2_001.fastq.gz # 50 bp: barcodes, UMI and linkers, poly-T
Sample02_S2_R1_001.fastq.gz # 50 bp: cDNA reads
Sample02_S2_R2_001.fastq.gz # 50 bp: barcodes, UMI and linkers, poly-T
# V3 configuration
Sample01_S1_I1_001.fastq.gz # 6 bp: sample index, can be ignored
Sample01_S1_R1_001.fastq.gz # 50 bp: cDNA reads
Sample01_S1_R2_001.fastq.gz # 8 bp: Barcode1
Sample01_S1_R3_001.fastq.gz # 14 bp: Barcode2 + UMI
Sample02_S2_I1_001.fastq.gz # 6 bp: sample index, can be ignored
Sample02_S2_R1_001.fastq.gz # 50 bp: cDNA reads
Sample02_S2_R2_001.fastq.gz # 8 bp: Barcode1
Sample01_S2_R3_001.fastq.gz # 14 bp: Barcode2 + UMI
For the V1 and V2 configurations, you are ready to go. However, since the cell barcodes and UMI are distributed in different reads, we need to collect them into one fastq file in order to use starsolo. This can be done by simple stitching the reads:
# Sample01
paste <(zcat Sample01_S1_R2_001.fastq.gz) \
<(zcat Sample01_S1_R3_001.fastq.gz) | \
awk -F '\t' '{ if(NR%4==1||NR%4==3) {print $1} else {print $1 $2} }' | \
gzip > Sample01_S1_CB_UMI.fastq.gz
# Sample02
paste <(zcat Sample02_S2_R2_001.fastq.gz) \
<(zcat Sample02_S2_R3_001.fastq.gz) | \
awk -F '\t' '{ if(NR%4==1||NR%4==3) {print $1} else {print $1 $2} }' | \
gzip > Sample02_S2_CB_UMI.fastq.gz
After that, you are ready to go.
Public Data#
For the purpose of demonstration, we will use the inDrop data from the following paper:
Note
Mereu E, Lafzi A, Moutinho C, Ziegenhain C, McCarthy DJ, Álvarez-Varela A, Batlle E, Sagar, Grün D, Lau JK, Boutet SC, Sanada C, Ooi A, Jones RC, Kaihara K, Brampton C, Talaga Y, Sasagawa Y, Tanaka K, Hayashi T, Braeuning C, Fischer C, Sauer S, Trefzer T, Conrad C, Adiconis X, Nguyen LT, Regev A, Levin JZ, Parekh S, Janjic A, Wange LE, Bagnoli JW, Enard W, Gut M, Sandberg R, Nikaido I, Gut I, Stegle O, Heyn H (2020) Benchmarking single-cell RNA-sequencing protocols for cell atlas projects. Nat Biotechnol 38:747–755. https://doi.org/10.1038/s41587-020-0469-4
where the authors benchmarked quite a few different scRNA-seq methods using a standardised sample: a mixture of different human, mouse and dog cells. We are going to use the data from the inDrop method, which uses the commercial 1CellBio platform. You can download the fastq file from this ENA page:
# get fastq files
mkdir -p mereu2020/indrop
wget -P mereu2020/indrop -c \
ftp://ftp.sra.ebi.ac.uk/vol1/fastq/SRR962/004/SRR9621794/SRR9621794_1.fastq.gz \
ftp://ftp.sra.ebi.ac.uk/vol1/fastq/SRR962/004/SRR9621794/SRR9621794_2.fastq.gz
Prepare Whitelist#
The full oligo sequences can be found in the Supplementary Table S2 and Supplementary Table S3 from the inDrop Nature Protocols paper. As you can see, there are a total of 384 different Barcode1 with 8 - 11 bp in length, and 384 different Barcode2. The oligos are added to the gel beads by primer extension during the split-pool procedures. The cell barcodes are basically the combination of Barcode1 and Barcode2. There will be a total of 384 * 384 = 147456 possible cell barcodes. I have organised the oligo information into two tables here (only showing 5 records):
Barcode1
Name |
Sequence |
Reverse complement |
|---|---|---|
W1-bc1.1-PE1 |
AAACAAAC |
GTTTGTTT |
W1-bc1.2-PE1 |
AAACACGGT |
ACCGTGTTT |
W1-bc1.3-PE1 |
AAACACTATC |
GATAGTGTTT |
W1-bc1.4-PE1 |
AAACCGCCTCA |
TGAGGCGGTTT |
Barcode2
Name |
Sequence |
Reverse complement |
|---|---|---|
BA19-N6-bc2.1-W1 |
AAACAAAC |
GTTTGTTT |
BA19-N6-bc2.2-W1 |
AAACACGG |
CCGTGTTT |
BA19-N6-bc2.3-W1 |
AAACACTA |
TAGTGTTT |
BA19-N6-bc2.4-W1 |
AAACCGCC |
GGCGGTTT |
You should notice that Barcode1 has variable lengths, but the first 8 bp are exactly the same as Barcode2. I have prepared the full tables in csv format for you to download:
inDrop_Barcode1.csv
inDrop_Barcode2.csv
Let’s download them to generate the whitelist:
wget -P mereu2020/indrop \
https://teichlab.github.io/scg_lib_structs/data/inDrop/inDrop_Barcode1.csv \
https://teichlab.github.io/scg_lib_structs/data/inDrop/inDrop_Barcode2.csv
Now we need to generate the whitelist of those two sets of barcodes. Read very carefully of the inDrop GitHub page. Pay attention to the oligo orientation. The barcode sequences that we get from the inDrop Nature Protocols paper are the sequences in the adaptors, which are used to generate the bead oligos. Therefore, the sequences on the bead oligos are reverse complement to the actual barcodes. Now, you can see that in the V1 and V2 configuration, Barcode1 and Barcode2 are in the same read and in the same direction of the bead oligo. Therefore, we should use the reverse complement of the barcode sequences for the whitelists. In the V3 configuration, Barcode1 is sequenced in the opposite direction of the bead oligo with only 8 cycles, so we need to use the first 8 bp of Barcode1 as they are. Barcode2 is sequenced in the same direction of the bead oligo, so we should take the reverse complement of the barcode sequence. In addition, since we stitch Barcode1, Barcode2 and UMI together into the CB_UMI.fastq.gz, we should generate all possible combinations of the Barcode1_8bp + Barcode2 rc as the whitelist. Here is how you could do this:
# for V1 and V2, prepare two plain lists from
# the reverse complement of Barcode1 and Barcode2
tail -n +2 mereu2020/indrop/inDrop_Barcode1.csv |
cut -f 3 -d, > mereu2020/indrop/V1and2_BC1.txt
tail -n +2 mereu2020/indrop/inDrop_Barcode2.csv |
cut -f 3 -d, > mereu2020/indrop/V1and2_BC2.txt
# for V3, we need to get all possible combination the first 8bp of the Barcode1
# to the reverse complement of Barcode2 sequence
for x in $(tail -n +2 mereu2020/indrop/inDrop_Barcode1.csv | cut -f 2 -d, | cut -c 1-8); do
for y in $(tail -n +2 mereu2020/indrop/inDrop_Barcode2.csv | cut -f 3 -d,); do
echo "${x}${y}"
done
done > mereu2020/indrop/V3_whitelist.txt
From FastQ To Count Matrix#
The public data was produced using the 1CellBio inDrop platform. Only have two fastq files are there. If you grep the sequence GAGTGATTGCTTGTGACGCCTT, it appears in Read 2 more often, so I assume it was based on V2. However, the perfect match only appears in ~24% of Read 2. I’m not entirely sure what’s going on, maybe the commercial 1CellBio platform has different mechanism. Anyway, let’s move on as if it is okay for the sake of demonstration.
# map and generate the count matrix
STAR --runThreadN 4 \
--genomeDir mix_hg38_mm10/star_index \
--readFilesCommand zcat \
--outFileNamePrefix mereu2020/star_outs/ \
--readFilesIn mereu2020/indrop/SRR9621794_1.fastq.gz mereu2020/indrop/SRR9621794_2.fastq.gz \
--soloType CB_UMI_Complex \
--soloAdapterSequence GAGTGATTGCTTGTGACGCCTT \
--soloAdapterMismatchesNmax 4 \
--soloCBposition 0_0_2_-1 3_1_3_8 \
--soloUMIposition 3_9_3_14 \
--soloCBwhitelist mereu2020/indrop/V1and2_BC1.txt mereu2020/indrop/V1and2_BC2.txt \
--soloCBmatchWLtype 1MM \
--soloCellFilter EmptyDrops_CR \
--soloStrand Forward \
--outSAMattributes CB UB \
--outSAMtype BAM SortedByCoordinate
Explanation#
If you understand the inDrop experimental procedures described in this GitHub Page, the command above should be straightforward to understand.
--runThreadN 4
Use 4 cores for the preprocessing. Change accordingly if using more or less cores.
--genomeDir mix_hg38_mm10/star_index
Pointing to the directory of the star index. The public data from the above paper was produced using the HCA reference sample, which consists of human PBMCs (60%), and HEK293T (6%), mouse colon (30%), NIH3T3 (3%) and dog MDCK cells (1%). Therefore, we need to use the species mixing reference genome. We also need to add the dog genome, but the dog cells only take 1% of all cells, so I did not bother in this documentation.
--readFilesCommand zcat
Since the
fastqfiles are in.gzformat, we need thezcatcommand to extract them on the fly.
--outFileNamePrefix mereu2020/star_outs/
We want to keep everything organised. This parameter directs all output files into the
mereu2020/star_outs/directory.
--readFilesIn
If you check the manual, we should put two files here. The first file is the reads that come from cDNA, and the second file should contain cell barcode and UMI. In inDrop V2, cDNA reads come from Read 1, and the cell barcode and UMI come from Read 2. Check the inDrop GitHub Page if you are not sure.
--soloType CB_UMI_Complex
Since Read 2 not only has cell barcodes and UMI, the common linker sequences are also there. The cell barcodes are non-consecutive, separated by the linker sequences. In this case, we have to use the
CB_UMI_Complexoption. Of course, we could also extract them upfront into a newfastqfile, but that’s slow. It is better to use this option.
--soloAdapterSequence GAGTGATTGCTTGTGACGCCTT
The 8 - 11 bp variable length of Barcode1 at the beginning of Read 2 makes the situation complicated, because the absolute positions of Barcode2 and UMI in each read will vary. However, by specifying an adaptor sequence, we could use this sequence as an anchor, and tell the program where cell barcodes and UMI are located relatively to the anchor. See below.
--soloAdapterMismatchesNmax 3
The number of mismatches are tolerated during the adapter finding. The adapter here is a bit long, so I want a bit relaxed matching, but you may want to try a few different options, like 1 (the default) or 2.
--soloCBposition and --soloUMIposition
These options specify the locations of cell barcode and UMI in the 2nd fastq files we passed to
--readFilesIn. In this case, it is Read 2. Read the STAR manual for more details. I have drawn a picture to help myself decide the exact parameters. There are some freedom here depending on what you are using as anchors. in inDrop V1 & 2, the Barcode1 has variable lengths, the absolute positions of Barcode2 and UMI are variable. Therefore, using Read start as anchor will not work for them. We need to use the adaptor as the anchor, and specify the positions relative to the anchor. See the image:
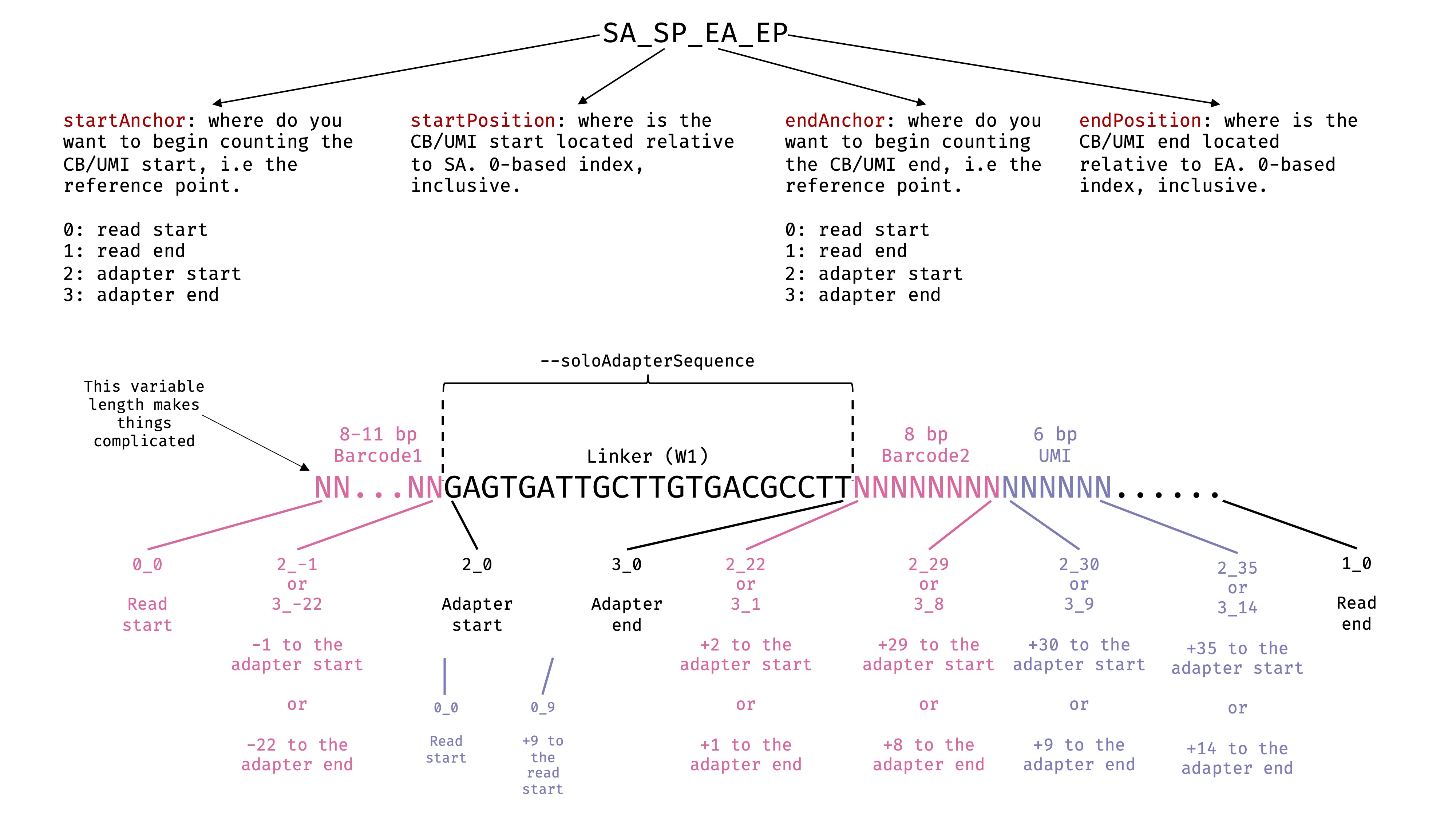
--soloCBwhitelist
Since the real cell barcodes consists of two non-consecutive parts: two sets of barcodes. The whitelist here is the combination of those two lists. We should provide them separately in the specified order and
starwill take care of the combinations.
--soloCBmatchWLtype 1MM
How stringent we want the cell barcode reads to match the whitelist. The default option (
1MM_Multi) does not work here. We choose this one here for simplicity, but you might want to experimenting different parameters to see what the difference is.
--soloCellFilter EmptyDrops_CR
Experiments are never perfect. Even for barcodes that do not capture the molecules inside the cells, you may still get some reads due to various reasons, such as ambient RNA or DNA and leakage. In general, the number of reads from those cell barcodes should be much smaller, often orders of magnitude smaller, than those barcodes that come from real cells. In order to identify true cells from the background, you can apply different algorithms. Check the
starmanual for more information. We useEmptyDrops_CRwhich is the most frequently used parameter.
--soloStrand Forward
The choice of this parameter depends on where the cDNA reads come from, i.e. the reads from the first file passed to
--readFilesIn. You need to check the experimental protocol. If the cDNA reads are from the same strand as the mRNA (the coding strand), this parameter will beForward(this is the default). If they are from the opposite strand as the mRNA, which is often called the first strand, this parameter will beReverse. In the case of inDrop V2, the cDNA reads are from the Read 1 file. During the experiment, the mRNA molecules are captured by barcoded oligo-dT primer containing UMI and Read 2 sequencing primer. Therefore, Read 2 consists of cell barcodes and UMI. They come from the first strand, complementary to the coding strand. Read 1 comes from the coding strand. Therefore, useForwardfor inDrop V2 data. ThisForwardparameter is the default, because many protocols generate data like this, but I still specified it here to make it clear. Check the inDrop GitHub Page if you are not sure.
--outSAMattributes CB UB
We want the cell barcode and UMI sequences in the
CBandUBattributes of the output, respectively. The information will be very helpful for downstream analysis.
--outSAMtype BAM SortedByCoordinate
We want sorted
BAMfor easy handling by other programs.
If everything goes well, your directory should look the same as the following:
scg_prep_test/mereu2020/
├── indrop
│ ├── inDrop_Barcode1.csv
│ ├── inDrop_Barcode2.csv
│ ├── SRR9621794_1.fastq.gz
│ ├── SRR9621794_2.fastq.gz
│ ├── V1and2_BC1.txt
│ ├── V1and2_BC2.txt
│ └── V3_whitelist.txt
└── star_outs
├── Aligned.sortedByCoord.out.bam
├── Log.final.out
├── Log.out
├── Log.progress.out
├── SJ.out.tab
└── Solo.out
├── Barcodes.stats
└── Gene
├── Features.stats
├── filtered
│ ├── barcodes.tsv
│ ├── features.tsv
│ └── matrix.mtx
├── raw
│ ├── barcodes.tsv
│ ├── features.tsv
│ └── matrix.mtx
├── Summary.csv
└── UMIperCellSorted.txt
6 directories, 22 files
Some Extra Work#
If you check the content of the mereu2020/star_outs/Solo.out/Gene/Summary.csv file, you will see that only ~24% of reads have valid barcodes. The commercial platform is difficult to crack. Let’s use the sample data provided by the indrops GitHub repository to see if the above procedures we build are good or not.
# first, clone the indrops github
git clone https://github.com/indrops/indrops.git
# process V1 data, remember Read 2 is the cDNA in V1
STAR --runThreadN 4 \
--genomeDir mix_hg38_mm10/star_index \
--readFilesCommand zcat \
--outFileNamePrefix indrops/v1_outs/ \
--readFilesIn indrops/test/seq_runs/run_v1_single_file/R2.fastq.gz indrops/test/seq_runs/run_v1_single_file/R1.fastq.gz \
--soloType CB_UMI_Complex \
--soloAdapterSequence GAGTGATTGCTTGTGACGCCTT \
--soloAdapterMismatchesNmax 3 \
--soloCBposition 0_0_2_-1 3_1_3_8 \
--soloUMIposition 3_9_3_14 \
--soloCBwhitelist mereu2020/indrop/V1and2_BC1.txt mereu2020/indrop/V1and2_BC2.txt \
--soloCBmatchWLtype 1MM \
--soloCellFilter EmptyDrops_CR \
--soloStrand Forward \
--outSAMattributes CB UB \
--outSAMtype BAM SortedByCoordinate
# process V2 data, remember Read 1 is the cDNA in V2
STAR --runThreadN 4 \
--genomeDir mix_hg38_mm10/star_index \
--readFilesCommand zcat \
--outFileNamePrefix indrops/v2_outs/ \
--readFilesIn indrops/test/seq_runs/run_v2_single_file/R1.fastq.gz indrops/test/seq_runs/run_v2_single_file/R2.fastq.gz \
--soloType CB_UMI_Complex \
--soloAdapterSequence GAGTGATTGCTTGTGACGCCTT \
--soloAdapterMismatchesNmax 3 \
--soloCBposition 0_0_2_-1 3_1_3_8 \
--soloUMIposition 3_9_3_14 \
--soloCBwhitelist mereu2020/indrop/V1and2_BC1.txt mereu2020/indrop/V1and2_BC2.txt \
--soloCBmatchWLtype 1MM \
--soloCellFilter EmptyDrops_CR \
--soloStrand Forward \
--outSAMattributes CB UB \
--outSAMtype BAM SortedByCoordinate
If you check the Summary.csv files, more than 80% of reads contain valid barcodes. We know we are good.
In terms of V3, the file names are different from what we described at the beginning of the documentation. There are four types of fastq files in the repository directory indrops/test/seq_runs/run_v3. To put them into context in this documentation:
Files in the indrops repo |
Files in this documentation |
Description |
|---|---|---|
Undetermined_S0_R1_001.fastq.gz |
R1_001.fastq.gz |
cDNA reads |
Undetermined_S0_R2_001.fastq.gz |
R2_001.fastq.gz |
Barcode1 |
Undetermined_S0_R3_001.fastq.gz |
I1_001.fastq.gz |
Sample index, ignored! |
Undetermined_S0_R4_001.fastq.gz |
R3_001.fastq.gz |
Barcode2 + UMI |
# the files are split by lanes, so let's merge them
cat indrops/test/seq_runs/run_v3/*_R1_001.fastq.gz > indrops/test/seq_runs/run_v3/cDNA.fastq.gz
# stitch Barcode1 and Barcode2+UMI file
paste <(zcat indrops/test/seq_runs/run_v3/*_R2_001.fastq.gz) \
<(zcat indrops/test/seq_runs/run_v3/*_R3_001.fastq.gz) | \
awk -F '\t' '{ if(NR%4==1||NR%4==3) {print $1} else {print $1 $2} }' | \
gzip > indrops/test/seq_runs/run_v3/CB_UMI.fastq.gz
# do the mapping
# Note R4 contains some extra T after Barcode2+UMI
# so we need --soloBarcodeReadLength 0 to turn off length check
STAR --runThreadN 4 \
--genomeDir mix_hg38_mm10/star_index \
--readFilesCommand zcat \
--outFileNamePrefix indrops/v3_outs/ \
--readFilesIn indrops/test/seq_runs/run_v3/cDNA.fastq.gz indrops/test/seq_runs/run_v3/CB_UMI.fastq.gz \
--soloType CB_UMI_Simple \
--soloCBstart 1 --soloCBlen 16 --soloUMIstart 17 --soloUMIlen 6 \
--soloBarcodeReadLength 0 \
--soloCBwhitelist mereu2020/indrop/V3_whitelist.txt \
--soloCellFilter EmptyDrops_CR \
--soloStrand Forward \
--outSAMattributes CB UB \
--outSAMtype BAM SortedByCoordinate
If you check the Summary.csv, it seems >60% reads have valid barcodes. This might be okay, but I expect to have higher fraction.